RNA Transcription/processing
Transcriptional regulation
As usual, images from wikicommons.
The first question is, how does the machine that makes RNA recognize a “gene?” In the past, I’ve called a “gene” the stretch of DNA encoding a protein and its regulatory sequences. We can separate it into the “structural gene,” which contains the actual code of the protein as it would be found in the mRNA, and the regulatory sequences. A very basic regulatory region is called the “promotor.” It is a region that serves as the initial recognition site that says: “This is a gene, transcribe here.”
In addition to the hydrogen bonds that make up the “watson-crick” base pairs, there are additional sites for hydrogen bonds and other contacts that can be made without “unzipping” the DNA, primarily along the major groove. In particular, the major grove is just about the right size for an alpha helix of protein to fit in. There, R-chains on the outside of the helix can make specific contacts with the bases and “read” the sequence (that is, bind in a sequence-specific manner). The first step in transcription is recognition of the promotor, almost always upstream from the structural gene. There are several types of DNA binding proteins. Here are two examples:
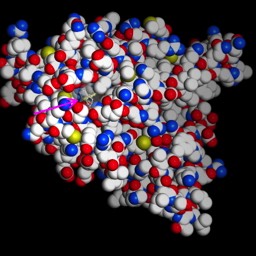
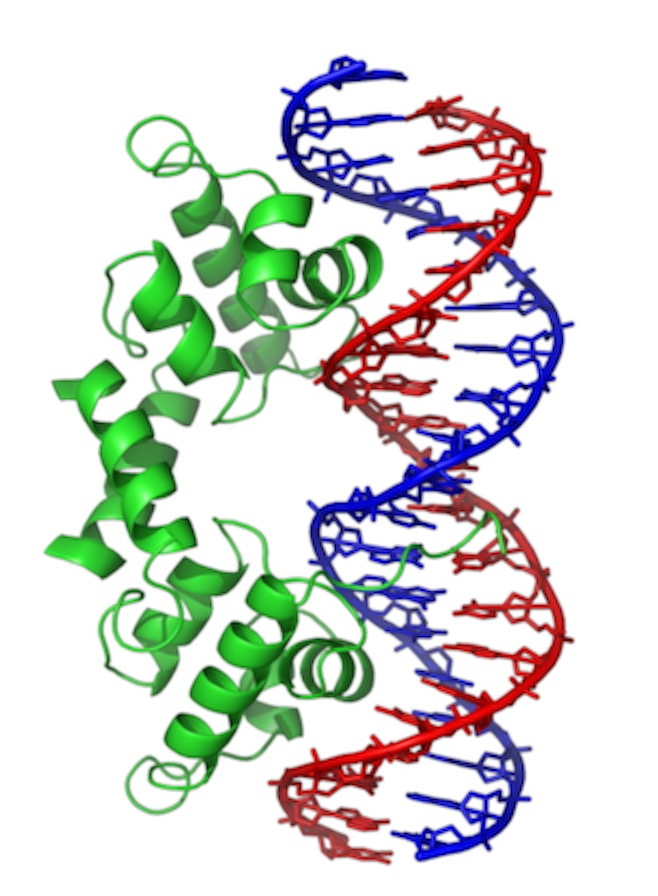 This is “Lambda repressor.” Notice two things: the protein binds as a dimer. In fact, each is identical and the overall DNA sequence is a “palindrome.” More on that later. Second, notice that one of the alpha-helices in each subunit is reaching down into the major groove, where it makes sequence specific contacts. This particular protein actually blocks access of the RNA polymerase in E. coli. But, similar proteins act to promote binding.
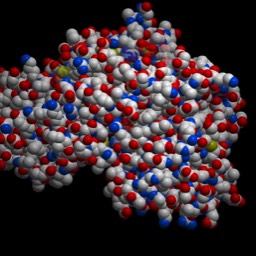
This is “Lambda repressor.” Notice two things: the protein binds as a dimer. In fact, each is identical and the overall DNA sequence is a “palindrome.” More on that later. Second, notice that one of the alpha-helices in each subunit is reaching down into the major groove, where it makes sequence specific contacts. This particular protein actually blocks access of the RNA polymerase in E. coli. But, similar proteins act to promote binding. 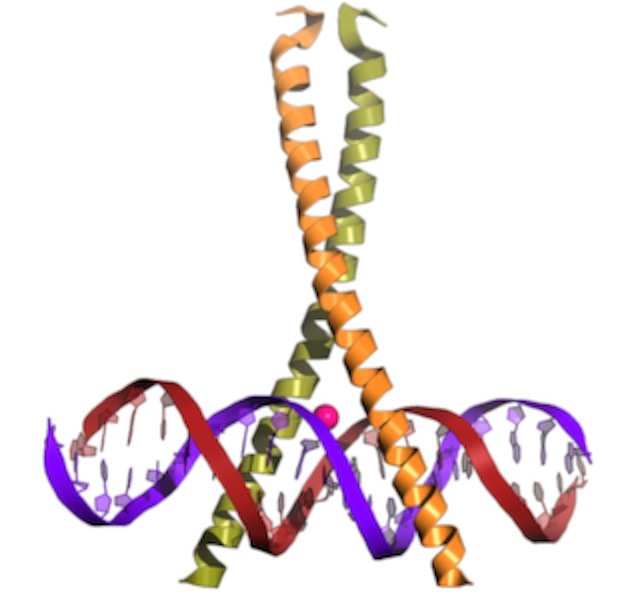 This is a b-zip protein binding domain, of a class that acts to promote transcription in our cells. Notice the same two things. Even though the overall structure of the proteins is different (note that much of the structure is left out), the binding has some similarities.
This is a b-zip protein binding domain, of a class that acts to promote transcription in our cells. Notice the same two things. Even though the overall structure of the proteins is different (note that much of the structure is left out), the binding has some similarities.Assembly of the transcription complex
What is the sequence?
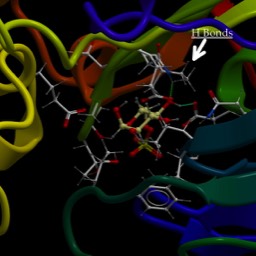
Well, that depends. The promotor is the most important thing that determines what genes are expressed (turned on) in which cells. Thus, genes that have to be on in every cell have different promotors than those only on in some. Genes that are turned on following a signal cascade have specific promotors. One of the main “housekeeping” promotors has a sequence called the “TATA box” (I wonder if you can guess why. It is bound by a complex depicted below. This is known as a “General Transcription factor” because it is common to most genes.
As you can see, the "Tata Binding Protein," or TBP interacts with the DNA over a larger region than just the TATA box. It also partially opens the DNA, where it bends it rather strongly. You may remember from the video we saw that there was a sharp bend to the DNA where the initiation complex was forming. It's also a little unusual in that it binds to the minor groove, using a beta-sheet domain. That bends and partially opens the DNA.
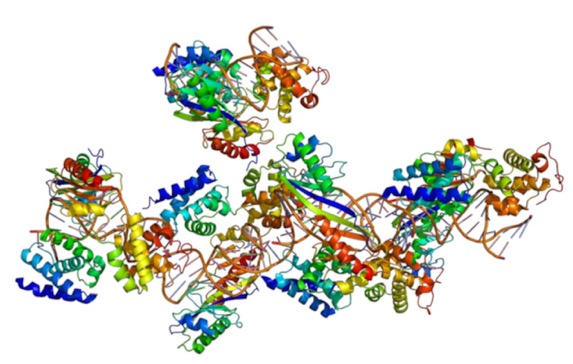
After initial proteins assemble at the binding site, they serve as binding sites for still more proteins that will be necessary to start transcription.
Here is a depiction of some of the proteins involved:
Notice the factors off to the right labeled 4 and 5. These have more to do with those larger levels of regulation I was talking about.
Here is a cool video on the process that shows some of the next step also:
RNA Splicing part 1
Take a look at this video which is presented by Cold Spring Harbor Lab, where I used to work years ago and where some of the work I discussed today was done.
The parts of the mRNA that are removed and discarded are called "introns," while the parts that are maintained in the final, mature mRNA are called exons. The exons include the portion that codes for the protein, as well as other regulatory sequences both before and after the "coding" region. As the video describes, there are complexes of proteins and RNA that do the splicing. The whole complex is called the "Spliceosome." The components include the generically named "small nuclear RNAs" which assemble with proteins to form "Small Nuclear Ribonuclear Proteins," or "snurps."
The story of how and why this happens is one of my favorite topics. We will discuss this more later.
RNA Processing
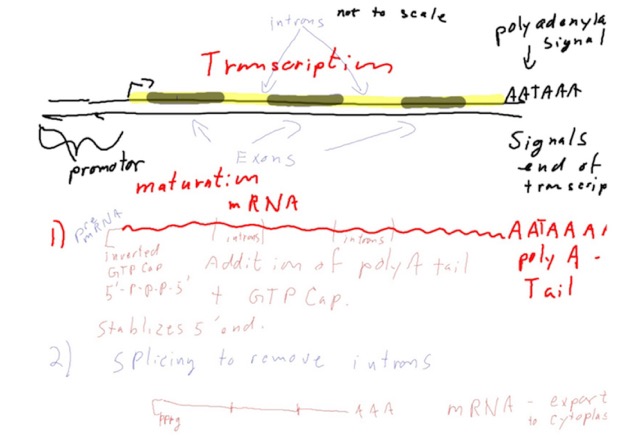 All Most of what I’ll be talking about here occurs specifically in eukaryotes.
All Most of what I’ll be talking about here occurs specifically in eukaryotes. As usual, most images are from Wikicommons.
Following transcription, the “pre-mRNA” must be processed on its way out of the nucleus to the cytoplasm.
First, notice that the sequence that made up the promotor does not end up in the pre-mRNA. This is a general theme: With each step in the DNA→RNA→Protein pathway, the information needed to specify regulation of each step generally is lost as we go to the next step. This makes sense, since it is no longer needed. Jerry had the clever realization of that fact today in class.
Poly-A tail and GTP cap.
This addresses two issues: what is the "stop transcription signal" and how do we protect both ends of the mRNA from being rapidly broken down?
When the RNA polymerase reaches the end of the sequence that’s supposed to be transcribed, it hits a signal called the “polyadenylation signal.” This is the "stop transcription signal," but also results in something else happening. The sequence in the DNA (coding strand) is 5'-AATAAA. Of course, the polymerase is reading the template strand, so you could say the “signal” really is 3' TTTATT. The sequence varies a fair bit. This signals the RNA polymerase to leave the DNA. The sequence now in the pre mRNA (AAUAAA, or something similar) recruits a protein complex that will cleave the mRNA near the 3' end and an enzyme called PAP, for Poly Adenyl Polymerase, uses ATP to add a long series of A’s to the end of the mRNA. These A’s, and there may be hundreds of them, are NOT encoded anywhere. This is the reason there was a 3' extension at the end of the mRNA in the picture today. To be fair, the researchers already knew about this and it is part of how they oriented themselves on the DNA/RNA duplex).
The utility of the tail seems to be in recruiting several poly-A binding proteins that protect the mRNA from nucleases that degrade it, facilitate the next steps in processing, the transport of the mRNA out of the nucleus to the cytoplasm, and regulation of translation.
Some prokaryotes do a version of poly-A tail also. But, again, most of this applies to eukaryotes.
The other initial change is the addition of the GTP cap. Actually, it’s a modified G with a methyl group on position 7 of the base, cleverly called 7-methyl GTP. Wiki has a short discussion of the mechanism of transfer. There is a specific enzyme that does the capping and the cap looks something like this:
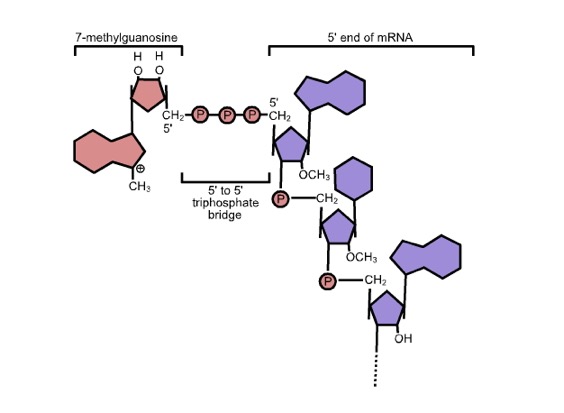
Note the “inverted” 5'-5' link. The cap structure also protects the end of the mRNA from degradation.
RNA splicing:
The final processed mRNA might look something like this:

It comprises the coding sequence, the cap, the 5' and 3' “untranslated region” (UTR) and the poly-A tail. I think all the names are pretty self-explanatory. The 5' UTR, in particular, will contain sequences that contribute to regulating translation. Only the green stretch above will make it into the protein.
RNA Splicing part more detail
Two benefits of splicing
While no one believes that RNA splicing evolved because of these benefits, these are real benefits organisms now enjoy because of it.
- Alternative splicing: Not all the exons are included when splicing of specific mRNAs takes place. Different versions of the mRNA may be formed. In the example I gave earlier, exons 1, 3, and 4 could all be joined up in sequence, or, all 4 could be linked. This results in different versions of the protein, with different functions. The domain structure of proteins makes this possible. NOTE: you do not alter the order of the exons when this is done
- Exon shuffling: Also because of the domain structure of proteins, it is possible to add exons to genes via recombination and create different versions of the protein. Any chunk of DNA that has portions of introns on its ends that then is recombined (at the DNA level) into another intron will do no damage to an existing gene (It can be spliced out at the mRNA level). But, it also provides the possibility, through alternative splicing, to evolve proteins with new functions.
Spliceosome: (there’s a good descriptive name) It’s the assembly of proteins and RNA that carry out splicing.
snRNPs (pronounced “snurps”). These are “small nuclear ribonuclear proteins.” They are the components of the spliceosome.
snRNAs: small nuclear RNAs are the main catalytic components. It is the RNA that carries out the reaction. They have names like “U1,” and “U2.” etc.
Self Splicing:
The story about how RNAs got spliced became more obvious when self-splicing RNAs were found. I mentioned these today.
The general reaction looks like this:
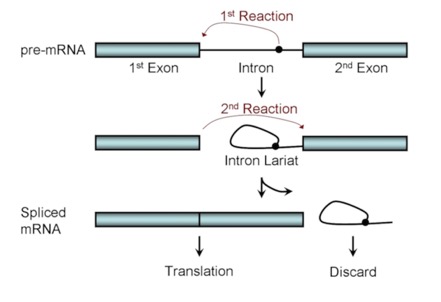
There are two, successive trans esterification reactions that occur in a concerted way and use energy from an additional GTP that is brought in by the complex. The first takes a branch point sequence near the 3' end of the intron. The 2' OH of that branch point attacks the 5' exon/intron boundary. This splices the intron into a lariat and frees the 3' end of the exon, which then attack the 5' end of the next exon, resulting in the spliced exons and a lariat structure of the intron.
In the self-splicing form, “internal guide sequences” form base-paired structures that hold the players together and facilitate the attacks (which usually involve that 2' OH in an intermediate).
You can see below how the stem-loop structures formed using base pairs, then can fold into three-dimensional structures, facilitating catalysis. Yes, I know, it should have been obvious.
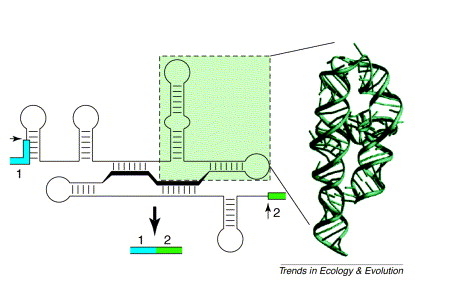
You can see how the base pairing can be used to bring the active sites together in more detail here. By the way, these last two images are used without permission. The one below is from the Molecule of the Month blog/discussion and the one above is from Trends in Ecology and Evolution.
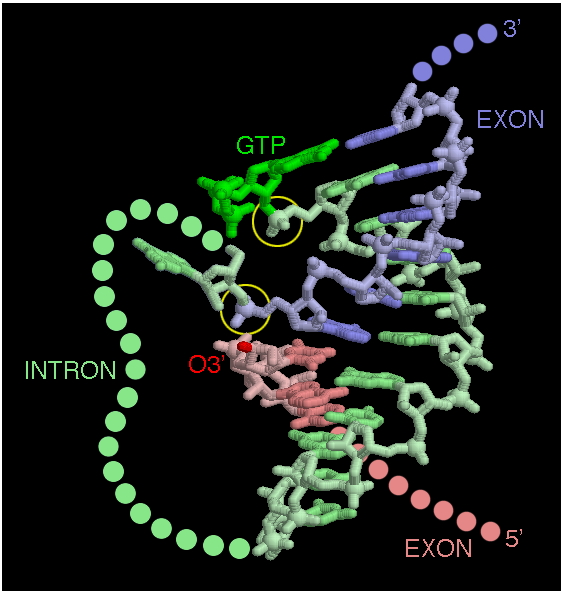
Normal mRNA splicing (non self-splicing)
The story is not all that different in the splicing of mRNA. There are guide sequences and a branch-point lariat. The chemistry varies a little as to who attacks whom when and where. But, the main difference is that the sequence of the intron is not that important for the structure to work. Instead of the intron folding into a complex structure, the separate RNAs in the snRNPs form those structures and then bind the specific sequences in the intron and at the intron-exon borders.