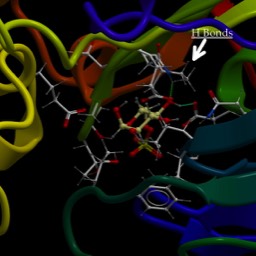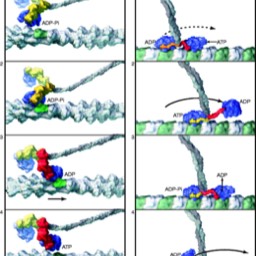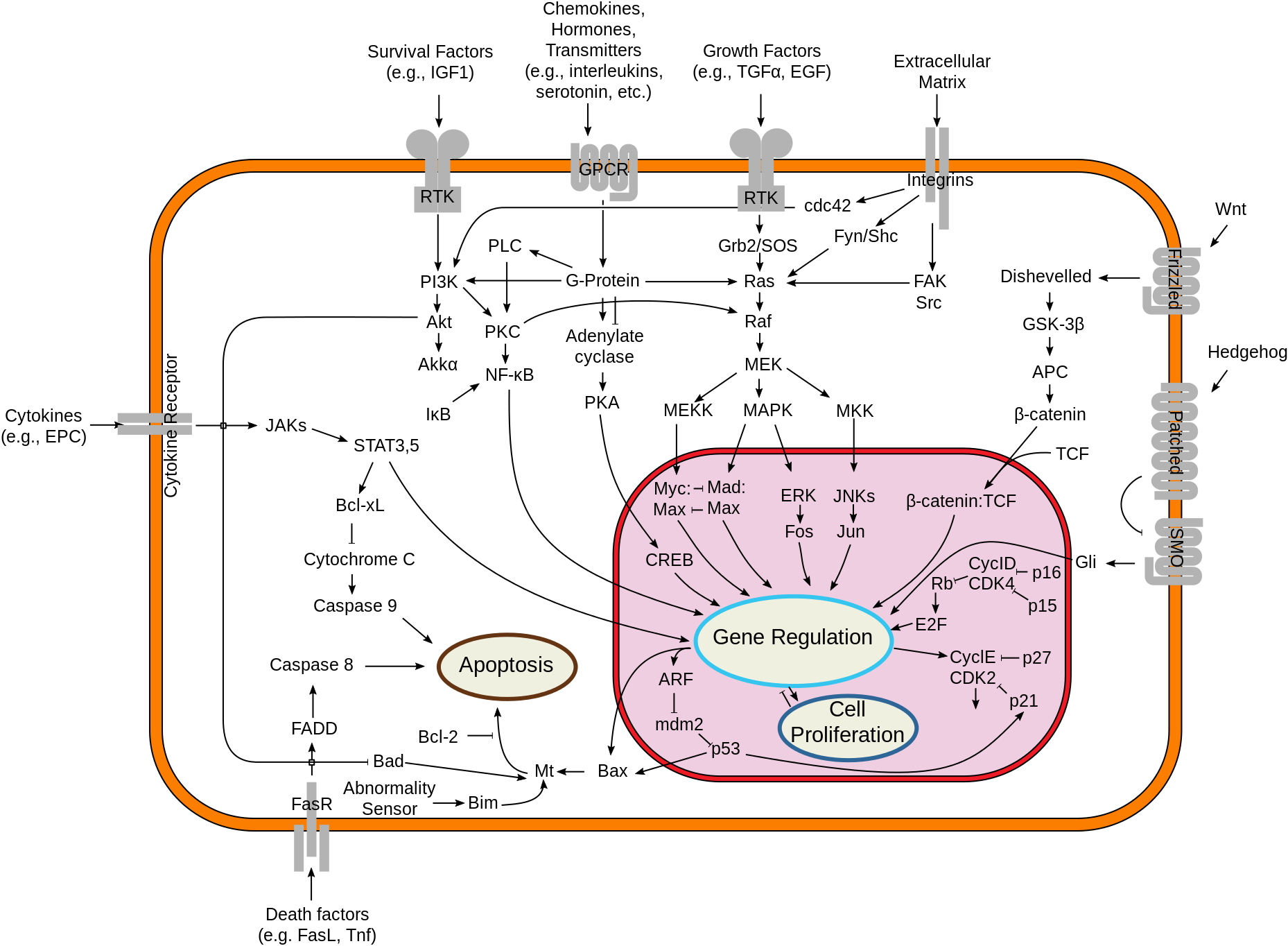
Signaling Introduction
This is the simplest image of cell signaling that has enough information to be useful. Start by looking at the types of signals that get to the cell. Ligands (growth factors...survival factors...death signals) all work by binding to cell-surface receptors. Look at the type of receptors available and the way the messages leave the membrane area. Look up “Receptor-tyrosine kinase” either in the book or online just to pick an example of how a signal cascade starts off.
Cell communication takes many forms, but there are commonalities in most of them. Cells may communicate by direct contacts between proteins on their surface, but do not need to be in physical contact for most events. There are lots of examples of "distinctions without a difference." What I mean by that is that you will hear reference to endocrine or hormone signaling (signal molecule made in a gland and secreted into blood), autocrine signaling (signal molecule made by same cell receiving it), paracrine signaling (signal molecule made by cells nearby the receiver), neurotransmitters, cytokines, chemokines…there really is no difference in how any of these work. It's like asking "Did you drive in your car to get milk or walk to the store?" Sure…there are differences…but the milk is the same.
In general, there are three steps:
We have looked at a small subset of G-protein-coupled receptors. These are among the oldest and most diverse types of signal molecules. We won’t look at all the possibilities, just one or two.
There is a decent animation of G-protein signaling here. The structural representation is not so good. But, it’s not too bad.
Here is a link to David's "molecule of the month" blog on the beta adrenergic receptor.
The key points are that:
In addition, if you have time, there is a wonderful lecture by Bob Lefkowitz, the discoverer of the beta andrenergic receptor (and the first GPCR other than rhodopsin) that can be found here. It’s long (50 minutes, but may be worth class time to see and discuss). He does a great job of discussing out the experiments to show how it works were done.
You may notice that there are many other animations for G-protein coupled signaling at You Tube. Some of them open ion channels (as in nerve cells), some of them connect to adenyl cyclase, as we have discussed. Others connect to other pathways, such as the phospholipase pathway.
The cAMP is the “second” messenger that goes off and activates many other proteins in the cell. Because each cell will have its own cAMP-responsive proteins, each cell may respond differently to the same signal molecule. As we discussed, there also will be, potentially, more than one receptor for a given ligand, resulting in different pathways being induced.
How is the signal turned off? Well, ligand may leave the receptor..but that doesn’t stop everything that is happening away from the receptor. The G-protein is still active, as is the adenyl cyclase and there is a lot of the second messenger, cAMP still floating around. The G-protein turns itself off by hydrolyzing the GTP back to GDP and lets go of its target protein (in this case, the adenyl cyclase) That turns off the target protein. Well…you still have all that cAMP around.
There is another enzyme that destroys that, converting it to AMP (not cyclic anymore). That enzyme is phosphodiesterase, often abbreviated PDE.
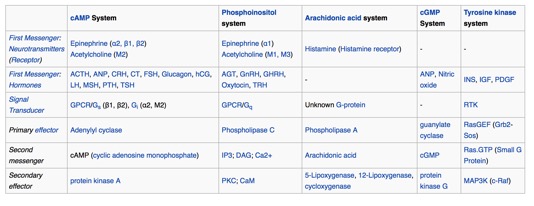
Below is a list of common first and
"second messengers." You may run into some of them.

- Here is a link to the signal talk I gave a while back, which includes some discussion on G proteins.
Overview:
Cell communication takes many forms, but there are commonalities in most of them. Cells may communicate by direct contacts between proteins on their surface, but do not need to be in physical contact for most events. There are lots of examples of "distinctions without a difference." What I mean by that is that you will hear reference to endocrine or hormone signaling (signal molecule made in a gland and secreted into blood), autocrine signaling (signal molecule made by same cell receiving it), paracrine signaling (signal molecule made by cells nearby the receiver), neurotransmitters, cytokines, chemokines…there really is no difference in how any of these work. It's like asking "Did you drive in your car to get milk or walk to the store?" Sure…there are differences…but the milk is the same.
In general, there are three steps:
- A signal molecule, or “ligand,” usually soluble (but can be bound on another cell or on the extracellular matrix), floats up to the target cell. The signal molecule may be a small protein (such as insulin), or a much smaller molecule (such as epinephrine). It binds to the extracellular face of a receptor, a protein that spans the membrane of the target cell
- The receptor changes shape (conformation) when the ligand binds. This is no different in concept to the idea of allosteric changes to proteins we discussed when we talked about inhibitors of enzymes. The tertiary structure of the protein changes. These changes affect not only the outside face of the receptor, but the internal side as well. This is the “transduction” event. That’s how the information gets through the membrane.
- The change in shape on the inside of the receptor leads to some change in the biochemistry of the cell. It may make available a new binding site, or a new active site. But some change inside the cell happens and ultimately, there are changes to how the cell behaves. There is a nearly limitless set of possibilities. Frequently, some enzyme gets activated and makes something called a “second messenger.” In this way the signal is amplified and may have many effects in the cell.
- (so much for "three steps") Eventually, the cell has to stop responding. The pathway has to have a way to shut down. The ligand may release from the receptor, but that only stops new signal events. Enzymes that were activated need to be deactivated. Second messengers made need to be destroyed.
More specific
We have looked at a small subset of G-protein-coupled receptors. These are among the oldest and most diverse types of signal molecules. We won’t look at all the possibilities, just one or two.
There is a decent animation of G-protein signaling here. The structural representation is not so good. But, it’s not too bad.
Here is a link to David's "molecule of the month" blog on the beta adrenergic receptor.
The key points are that:
- G proteins are inactive when GDP is bound, active when GTP is bound.
- Turning them “on” requires exchanging the GDP (kicking it out) for a GTP (allowing it to enter). This exchange is facilitated by the receptor, once ligand is bound.
- G proteins are molecular timers. Once turned on, they will turn themselves off by hydrolyzing the GTP to GDP.
- While they are in the “on state,” they activate other target proteins, usually enzymes, which then generate second messengers.
In addition, if you have time, there is a wonderful lecture by Bob Lefkowitz, the discoverer of the beta andrenergic receptor (and the first GPCR other than rhodopsin) that can be found here. It’s long (50 minutes, but may be worth class time to see and discuss). He does a great job of discussing out the experiments to show how it works were done.
You may notice that there are many other animations for G-protein coupled signaling at You Tube. Some of them open ion channels (as in nerve cells), some of them connect to adenyl cyclase, as we have discussed. Others connect to other pathways, such as the phospholipase pathway.
The cAMP is the “second” messenger that goes off and activates many other proteins in the cell. Because each cell will have its own cAMP-responsive proteins, each cell may respond differently to the same signal molecule. As we discussed, there also will be, potentially, more than one receptor for a given ligand, resulting in different pathways being induced.
How is the signal turned off? Well, ligand may leave the receptor..but that doesn’t stop everything that is happening away from the receptor. The G-protein is still active, as is the adenyl cyclase and there is a lot of the second messenger, cAMP still floating around. The G-protein turns itself off by hydrolyzing the GTP back to GDP and lets go of its target protein (in this case, the adenyl cyclase) That turns off the target protein. Well…you still have all that cAMP around.
There is another enzyme that destroys that, converting it to AMP (not cyclic anymore). That enzyme is phosphodiesterase, often abbreviated PDE.
Below is a list of common first and
"second messengers." You may run into some of them.